AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Ca element configuration9/15/2023  The electronic configuration of Calcium will be 1s2 2s2 2p6 3s2 3p6 4s2. How do you write the electron configuration for Calcium? A well as beryllium and aluminium, and unlike the alkaline metals, it doesn’t cause skin-burns. The metal is trimorphic, harder than sodium, but softer than aluminium. The electronic configuration of Calcium will be 1s2 2s2 2p6 3s2 3p6 4s2. The chemical element Calcium (Ca), atomic number 20, is the fifth element and the third most abundant metal in the earth’s crust. What is the electronic configuration of Calcium 20? How can I draw electronic configuration of calcium in a shell. What is the boiling Point of Calcium in Kelvin?īoiling Point of Calcium in Kelvin is 1757 K. Melting Point of Calcium in Kelvin is 1115 K. What is the melting Point of Calcium in Kelvin? What is the boiling Point of Calcium?īoiling Point of Calcium is 1757 K. Calcium has 20 electrons out of which 2 valence electrons are present in the 4s2 outer orbitals of atom. How many valence electrons does a Calcium atom have?Ĭalcium has 2 valence electrons. What is the color of Calcium?Ĭalcium is of Silver color. It is located in group 2 and period 4 in the modern periodic table. Calcium is the 20 element on the periodic table. What is the position of Calcium in the Periodic Table?Ĭalcium is a chemical element with the symbol Ca and atomic number 20. 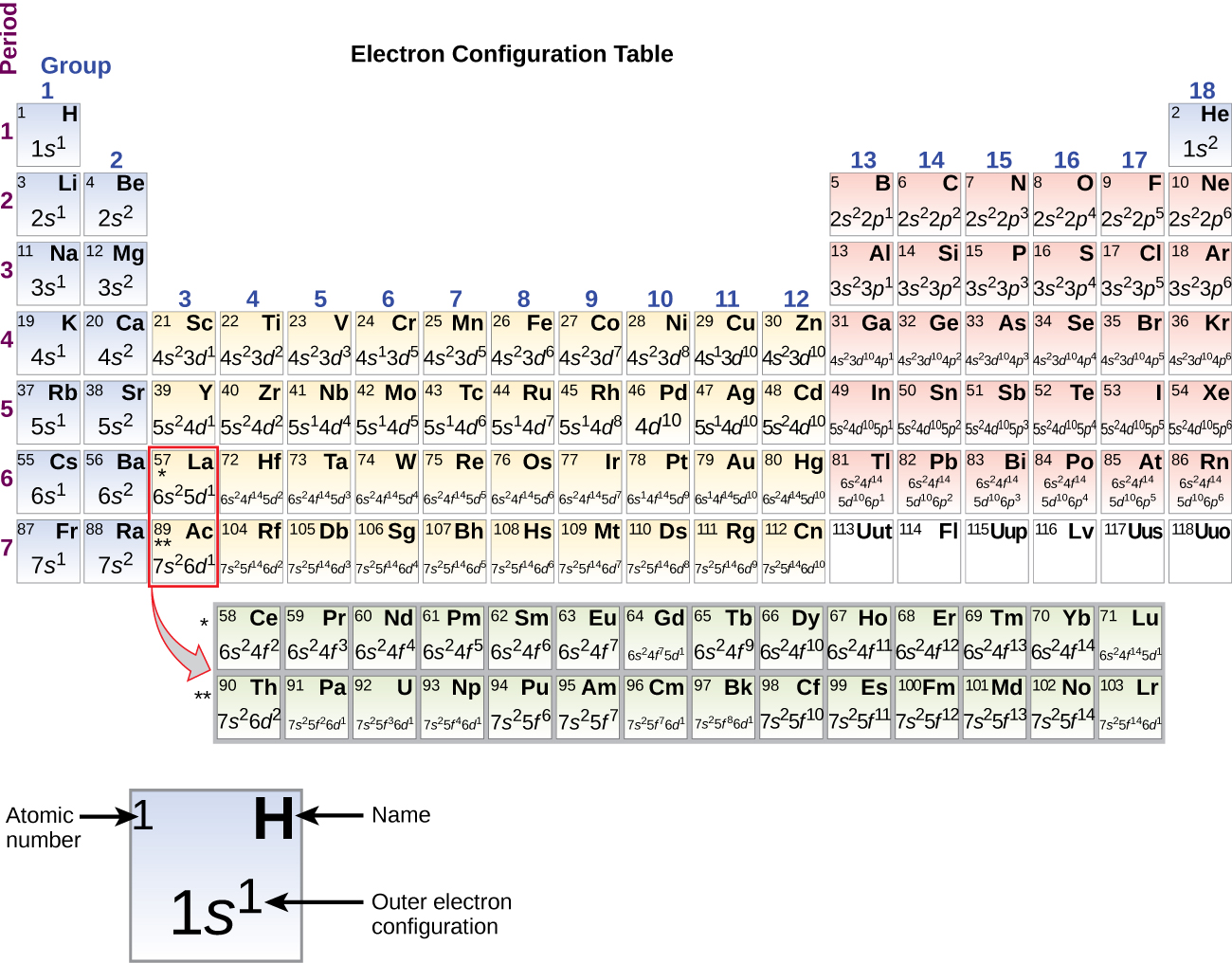
Calcium is a chemical element with symbol Ca and atomic number 20. To form abbreviated notation of electronic configuration, the completely filled subshells are replaced by the noble gas of the preceding period in square brackets. The abbreviated electronic configuration of Calcium is 4s2. With the help of the periodic table, we can easily see that the atomic number of calcium is 20. What is the abbreviated electronic configuration of Calcium? Calcium (Ca) Valence Electrons There are four simple steps to find out the valence electrons for calcium atom which are: Step 1: Find the Atomic Number To find out the atomic number of calcium, we can use the periodic table. The electronic configuration of Calcium is 1s2 2s2 2p6 3s2 3p6 4s2. What is the electronic configuration of Calcium? Optical Properties of Calcium Refractive IndexĪcoustic Properties of Calcium Speed of SoundĬalcium Thermal Properties - Enthalpies and thermodynamics Refer to table below for the Electrical properties ofCalcium Electrical ConductivityĬalcium Heat and Conduction Properties Thermal ConductivityĬalcium Magnetic Properties Magnetic Type Hardness of Calcium - Tests to Measure of Hardness of Element Mohs HardnessĬalcium is Conductor of electricity. Because its 2n shell is filled, it is energetically stable as a single atom and will rarely form chemical bonds with other atoms.Refer to below table for Calcium Physical Properties Densityġ.55 g/cm3(when liquid at m.p density is $1.378 g/cm3) For instance, lithium ( Li \text Ne start text, N, e, end text ), on the other hand, has a total of ten electrons: two are in its innermost 1 s 1s 1 s 1, s orbital and eight fill the second shell-two each in the 2 s 2s 2 s 2, s and three p p p p orbitals, 1 s 2 1s^ 2 1 s 2 1, s, squared 2 s 2 2s^ 2 2 s 2 2, s, squared 2 p 6 2p^6 2 p 6 2, p, start superscript, 6, end superscript. Elements in the second row of the periodic table place their electrons in the 2n shell as well as the 1n shell. After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. 
The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
